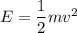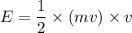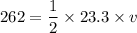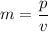Answers: 3


Another question on Physics

Physics, 22.06.2019 00:10
The energy released by a chemical reaction can be measured using a calorimeter. when barium hydroxide octahydrate crystals are reacted with dry ammonium chloride inside of a coffee cup calorimeter, the temperature of the 18.00 g of water in the calorimeter decreases from 30.0°c to 8.0°c. the equation for calculating energy absorbed or released by a reaction is: where q is the energy released or absorbed, m is the mass of water in the calorimeter, cp is the specific heat of water, and δt is the observed temperature change. if the specific heat of liquid water is 4.19 j/g·°c, how much energy was absorbed by the reaction?
Answers: 3

Physics, 22.06.2019 01:10
If vx = 7.00 units and vy = -7.60 units, determine the magnitude of v⃗ . determine the direction of v⃗ .
Answers: 2

Physics, 22.06.2019 04:30
Afeather of mass 0.001 kg falls from a height of 2 m. under realistic conditions, it experiences air resistance. based on what you know about friction, what can you say about the kinetic energy of the feather as it reaches the ground? acceleration due to gravity is g = 9.8 m/s2. a. ke < 0.0196 j b. ke = 00196 j c. je = 0 j d. ke > 0.0196 j
Answers: 1

You know the right answer?
An object is traveling such that it has a momentum of magnitude 23.3 kg. m/s and a kinetic energy of...
Questions

Geography, 05.05.2020 15:16

Mathematics, 05.05.2020 15:16

English, 05.05.2020 15:16

Mathematics, 05.05.2020 15:16

Mathematics, 05.05.2020 15:16

Mathematics, 05.05.2020 15:16

Mathematics, 05.05.2020 15:16

Mathematics, 05.05.2020 15:16



English, 05.05.2020 15:16


Mathematics, 05.05.2020 15:16


Social Studies, 05.05.2020 15:16


Mathematics, 05.05.2020 15:16


Mathematics, 05.05.2020 15:16

SAT, 05.05.2020 15:16