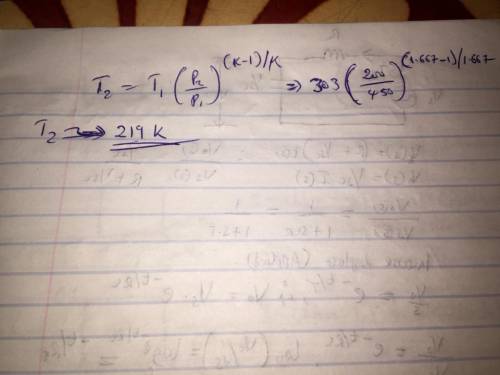Engineering, 02.07.2021 02:50 AliMe52
A well insulated rigid tank contains 4 kg of argon gas at 450 kPa and 30 C. A valve is opened, allowing the argon to escape until the tank pressure drops to 200 kPa. Assuming that the argon remaining in the tank experiences a reversible adiabatic process, find the final mass of argon in the tank. Since you don't have argon gas tables, assume cp, cv, k as needed at some appropriate temperature(s).

Answers: 3


Another question on Engineering

Engineering, 04.07.2019 18:20
Select any two (2) areas of applications of chain-drive. (clo4) a)-permanent lubrication necessary b)-hydraulic forklift truck operation c)-rigging and heavy moving materials d)-relatively high maintenance costs e)-costlier than belt drives
Answers: 2

Engineering, 04.07.2019 18:20
Steam enters a converging nozzle at 3.0 mpa and 500°c with a at 1.8 mpa. for a nozzle exit area of 32 cm2, determine the exit velocity, mass flow rate, and exit mach number if the nozzle: negligible velocity, and it exits (a) is isentropic (b) has an efficiency of 94 percent
Answers: 2


Engineering, 04.07.2019 19:10
For a process taking place in a closed system containing gas, the volume and pressure relationship is pvi-constant. -1.5 bar, the process starts with initial conditions, pi = =0.03 m3 and ends with final volume, v2-0.05 m3 determine the work done by the gas.
Answers: 2
You know the right answer?
A well insulated rigid tank contains 4 kg of argon gas at 450 kPa and 30 C. A valve is opened, allow...
Questions



Mathematics, 09.07.2019 03:30

Physics, 09.07.2019 03:30



Mathematics, 09.07.2019 03:30

Mathematics, 09.07.2019 03:30

Spanish, 09.07.2019 03:30

Mathematics, 09.07.2019 03:30

Chemistry, 09.07.2019 03:30

Mathematics, 09.07.2019 03:30

Mathematics, 09.07.2019 03:30



Mathematics, 09.07.2019 03:30

Mathematics, 09.07.2019 03:30

Biology, 09.07.2019 03:30