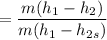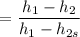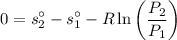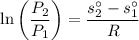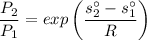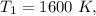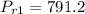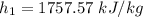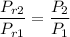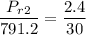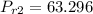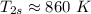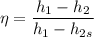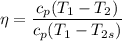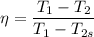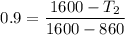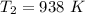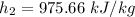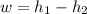Engineering, 30.03.2021 05:20 andersonrocksc
Air at 1600 K, 30 bar enters a turbine operating at steady state and expands adiabatically to the exit, where the pressure is 2.05 bar. The isentropic turbine efficiency is 90%. Assume ideal gas behavior for the air and ignore kinetic and potential energy effects. Determine the temperature at the exit, in K, and the work developed, in kJ per kg of air flowing.

Answers: 3


Another question on Engineering

Engineering, 03.07.2019 15:10
Two flowing streams of argon gas are adiabatically mixed to form a single flow/stream. one stream is 1.5 kg/s at 400 kpa and 200 c while the second stream is 2kg/s at 500 kpa and 100 ? . it is stated that the exit state of the mixed single flow of argon gas is 150 c and 300 kpa. assuming there is no work output or input during the mixing process, does this process violate either the first or the second law or both? explain and state all your assumptions.
Answers: 1

Engineering, 04.07.2019 18:10
Give heat transfer applications for the following, (i) gas turbines (propulsion) ) gas turbines (power generation). (iii) steam turbines. (iv) combined heat and power (chp). (v) automotive engines
Answers: 1

Engineering, 04.07.2019 18:10
Determine whether or not it is possible to compress air adiabatically from k to 140 kpa and 400 k. what is the entropy change during this process?
Answers: 3

Engineering, 04.07.2019 18:10
The thermal expansion or contraction of a given metal is a function of the f a)-density b)-initial temperature c)- temperature difference d)- linear coefficient of thermal expansion e)- final temperature f)- original length
Answers: 2
You know the right answer?
Air at 1600 K, 30 bar enters a turbine operating at steady state and expands adiabatically to the ex...
Questions






History, 06.07.2019 10:00

History, 06.07.2019 10:00









Physics, 06.07.2019 10:00



Biology, 06.07.2019 10:00

Spanish, 06.07.2019 10:00