
Engineering, 28.07.2020 16:01 wolfgirl10000
A small still is separating propane and butane at 135 °C, and initially contains 10 kg moles of a mixture whose composition is x = 0.3 (x = mole fraction butane). Additional mixture (x = 0.3) is fed at the rate of 5 kg mole/hr. The total volume of the liquid in the still is constant, and the concentration of the vapor from the still (xp) is related to x, as follows: Xp = How long will it take for X, to change from 0.3 to 0.35.

Answers: 3


Another question on Engineering

Engineering, 03.07.2019 14:10
Amass of m 1.5 kg of steam is contained in a closed rigid container. initially the pressure and temperature of the steam are: p 1.5 mpa and t 240°c (superheated state), respectively. then the temperature drops to t2= 100°c as the result of heat transfer to the surroundings. determine: a) quality of the steam at the end of the process, b) heat transfer with the surroundings. for: p1.5 mpa and t 240°c: enthalpy of superheated vapour is 2900 kj/kg, specific volume of superheated vapour is 0. 1483 m/kg, while for t 100°c: enthalpy of saturated liquid water is 419kj/kg, specific volume of saturated liquid water is 0.001043m/kg, enthalpy of saturated vapour is 2676 kj/kg, specific volume of saturated vapour is 1.672 m/kg and pressure is 0.1 mpa.
Answers: 3

Engineering, 04.07.2019 03:10
What precautions should you take to prevent injuries when dealing with heavy loads?
Answers: 1

Engineering, 04.07.2019 18:10
Fluids at rest possess no flow energy. a)- true b)- false
Answers: 3

Engineering, 04.07.2019 18:10
Compute the pressure drop of 30°c air flowing with a mean velocity of 8 m/s in a circular sheet-metal duct 300 mm in diameter and 15 m long. use a friction factor, f 0.02, and pair = 1.1644 kg/m a. 37.26 pa b. 25.27 pa n c. 29.34 pa d. 30.52 pa
Answers: 1
You know the right answer?
A small still is separating propane and butane at 135 °C, and initially contains 10 kg moles of a mi...
Questions

Health, 23.06.2019 11:30

Health, 23.06.2019 11:30

Mathematics, 23.06.2019 11:30

Mathematics, 23.06.2019 11:30

Health, 23.06.2019 11:30

Mathematics, 23.06.2019 11:30



Mathematics, 23.06.2019 11:30



Mathematics, 23.06.2019 11:30

Mathematics, 23.06.2019 11:30

History, 23.06.2019 11:30


Mathematics, 23.06.2019 11:30

Mathematics, 23.06.2019 11:30



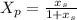 composition : propane = 0.70, butane = 0.3
composition : propane = 0.70, butane = 0.3 mol butane/mol condensed
mol butane/mol condensed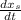 = 0.15 -
= 0.15 - 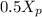
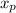
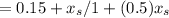
 = 0.3 and t = Ф,
= 0.3 and t = Ф,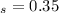
![[ - (x_{s} /0.35 - (1/(0.35)^2)* In(0.15 - 0.35x_{s} ) ]_{0.3} ^{0.35}](/tpl/images/0714/1297/a2c3f.png)


