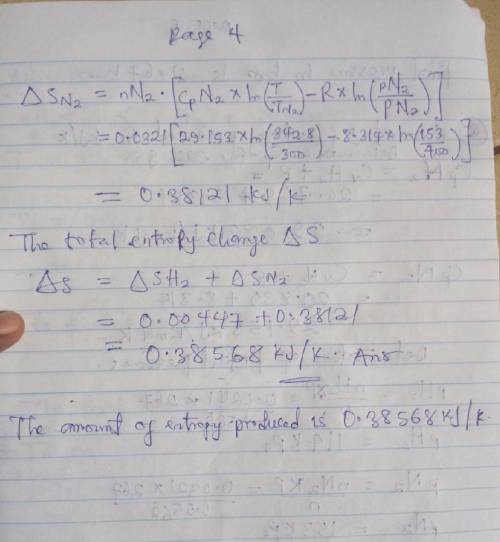Engineering, 30.03.2020 20:35 angel41vgg
An insulated tank having a total volume of 0.6 m3 is divided into two compartments. Initially one compartment contains 0.4 m3 of hydrogen at 127 oC, 2 bar and the other contains nitrogen at 27 oC, 4 bar. The gases are allowed to mix until an equilibrium state is attained. Assume the ideal gas model with constant specific heats, determine
a. The final temperature, in oC.
b. The final pressure, in bar.
c. The amount of entropy produced, in KJ/K.

Answers: 3


Another question on Engineering

Engineering, 03.07.2019 14:10
If the thermal strain developed in polyimide film during deposition is given as 0.0044. assume room temperature is kept at 17.3 c, and thermal coefficient of expansion for the film and the substrate are 54 x 10^-6c^-1 and 3.3 x 10^-6c^-1respectively. calculate the deposition temperature.
Answers: 3

Engineering, 04.07.2019 16:10
The force on a cutting tool are 2600n vertically downward and 2100 horizontal. determine the resultant force acting on the tool and the angle at which it acts.
Answers: 1

Engineering, 04.07.2019 18:10
Abrake has a normal braking torque of 2.8 kip in and heat-dissipating cast-iron surfaces whose mass is 40 lbm. suppose a load is brought to rest in 8.0 s from an initial angular speed of 1600 rev/min using the normal braking torque; estimate the temperature rise of the heat dissipating surfaces.
Answers: 3

Engineering, 04.07.2019 18:10
Manometers are good examples of measuring instruments, nowadays they are not as common as before. a)-capacitive probe gauges b)-gravitational gauges deformation ) gauges d)-digital gauges
Answers: 1
You know the right answer?
An insulated tank having a total volume of 0.6 m3 is divided into two compartments. Initially one co...
Questions

History, 26.10.2019 07:43


Biology, 26.10.2019 07:43



Biology, 26.10.2019 07:43

Biology, 26.10.2019 07:43






History, 26.10.2019 07:43

Social Studies, 26.10.2019 07:43

History, 26.10.2019 07:43


Business, 26.10.2019 07:43

English, 26.10.2019 07:43