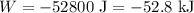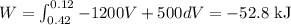Engineering, 18.02.2020 02:30 jwyapo4
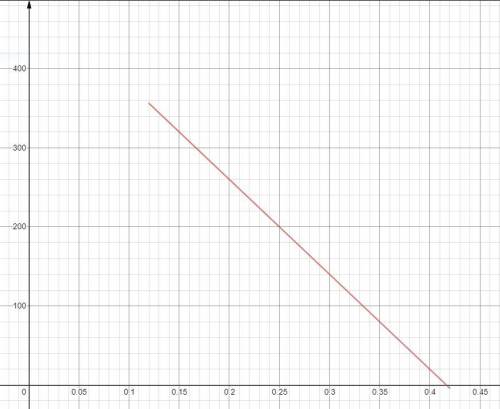
A gas is compressed from an initial volume of 0.42 m3 to a final volume of 0.12 m3. During the quasi-equilibrium process, the pressure changes with volume according to the relation P = aV + b, where a = –1200 kPa/m3 and b = 500 kPa. Calculate the work done during this process (a) by plotting the process on a P-V diagram and finding the area under the process curve and (b) by performing the necessary integrations.

Answers: 1


Another question on Engineering

Engineering, 04.07.2019 18:10
Afluid flows with a velocity field given by v=(x/t)i.. determine the local and convective accelerations when x=3 and t=1.
Answers: 2

Engineering, 04.07.2019 18:10
Which of the following controllers anticipates the future from the slope of errors over time? a)-proportional b)-on/off c)-integral d)-derivative.
Answers: 2

Engineering, 04.07.2019 18:20
Derive the correction factor formula for conical nozzle i=-(1+ cosa) and calculate the nozzle angle correction factor for a nozzle whose divergence hal-fangle is 13 (hint: assume that all the mass flow originates at the apex of the cone.
Answers: 3

Engineering, 04.07.2019 19:20
To save energy, the air supply to a 2000 ft office has been shut off overnight and the room temperature has dropped to 40°f. in the morning, the thermostat is reset to 70°f and warm air at 120 f begins to flov in at 200ft'/min. the air is well mixed within the room, and an equal mass flow of air at room temperature (changing with time) is withdrawn through a return duct. the air pressure is nearly 1 atm everywhere. ignoring heat transfer with the surroundings and kinetic and potential energy effects, estimate how long it takes for the room temperature to reach 70°f. plot the room temperature as a function of time.
Answers: 1
You know the right answer?
A gas is compressed from an initial volume of 0.42 m3 to a final volume of 0.12 m3. During the quasi...
Questions

Computers and Technology, 24.02.2020 18:09


Mathematics, 24.02.2020 18:09