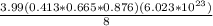Engineering, 18.01.2020 04:31 maymayrod2000
Ahypothetical metal has an orthorhombic unit cell for which the a, b, and c lattice parameters are 0.413 nm, 0.665 nm, and 0.876 nm, respectively. (a) if there are 8 atoms per unit cell and the atomic packing factor is 0.536, determine the atomic radius (in nm). (b) if the density is 3.99 g/cm3, calculate the metal's atomic weight (in g/mol).

Answers: 2


Another question on Engineering

Engineering, 04.07.2019 18:10
Thermal stresses are developed in a metal when its a) initial temperature is changed b) final temperature is changed c) density is changed d) thermal deformation is prevented e) expansion is prevented f) contraction is prevented
Answers: 2

Engineering, 04.07.2019 18:10
Burgers vector is generally parallel to the dislocation line. a)-true b)-false
Answers: 2

Engineering, 04.07.2019 18:10
The higher the astm grain size number, the finer the gran is. a)-true b)-false
Answers: 2

Engineering, 04.07.2019 18:20
Select any two (2) areas of applications of chain-drive. (clo4) a)-permanent lubrication necessary b)-hydraulic forklift truck operation c)-rigging and heavy moving materials d)-relatively high maintenance costs e)-costlier than belt drives
Answers: 2
You know the right answer?
Ahypothetical metal has an orthorhombic unit cell for which the a, b, and c lattice parameters are 0...
Questions



Geography, 08.11.2019 05:31


Mathematics, 08.11.2019 05:31

Biology, 08.11.2019 05:31


Health, 08.11.2019 05:31

Mathematics, 08.11.2019 05:31




Chemistry, 08.11.2019 05:31

Mathematics, 08.11.2019 05:31

English, 08.11.2019 05:31





English, 08.11.2019 05:31

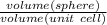 ..................1
..................1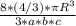
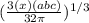
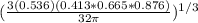
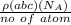 ....................2
....................2