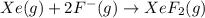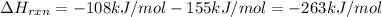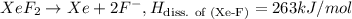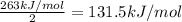Chemistry, 21.07.2019 07:30 Jessieileen
The enthalpy of formation of xef2(g) is –108 kj mol–1 and the bond dissociation enthalpy of the f–f bond is 155 kj mol–1 . what is the average bond dissociation enthalpy of a xe–f bond

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:30
Calculate the change in entropy if br2(l) is converted to br2(g). s° for br2(l) = 152.2 j/(mol•k) s° for br2(g) = 245.5 j/(mol•k) s° for br(g) = 175.0 j/(mol•k)
Answers: 3

Chemistry, 22.06.2019 03:00
Zoe is investigating the composition of substance a, an unknown substance. using chemical processes, she analyzes substance a and determines it is composed of sodium, oxygen, and hydrogen atoms in a ratio of 1 : 1 : 1. what is substance a? a. a compound b. an element c. a heterogeneous mixture d. a homogeneous mixture
Answers: 1

Chemistry, 22.06.2019 04:00
14. many depressants reduce small muscle control, making it harder for a. you to steer b. your mind to consider complex problems c. the eye to scan, focus, or stay still d. the kidneys to filter alcohol out of the bloodstream
Answers: 3

Chemistry, 22.06.2019 10:00
Ahydrogen atom has 1 electron. how many bonds can hydrogen form? a) 1 b) 2 c) 3 d) 4 e) 5
Answers: 3
You know the right answer?
The enthalpy of formation of xef2(g) is –108 kj mol–1 and the bond dissociation enthalpy of the f–f...
Questions



Social Studies, 08.10.2019 00:00













Geography, 08.10.2019 00:00




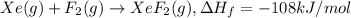 ..(1)
..(1)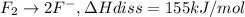 ..(2)
..(2)