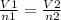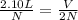Chemistry, 21.07.2019 09:00 itziabejar
If the number of moles of a gas initially contained in a 2.10 l vessel is doubled, what is the final volume of the gas in liters? (assume the pressure and temperature remain constant.) none of the above 8.40 4.20 1.05 6.30

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:20
Asolution is made by dissolving 25.5 grams of glucose (c6h12o6) in 398 grams of water. what is the freezing point depression of the solvent if the freezing point constant is -1.86 °c/m? show all of the work needed to solve this problem.
Answers: 1

Chemistry, 22.06.2019 02:30
List four observations that indicate that a chemical reaction may be taking place
Answers: 1

Chemistry, 22.06.2019 16:30
Asample of freon gas has a volume of 2.23 liters, a pressure of 4.85 kpa, and a temperature of -1.36°c. calculate the volume at a pressure of 1.38 kpa and a temperature of 5.5°c. (show work)
Answers: 1

Chemistry, 22.06.2019 19:30
To calculate percent by mass, use the equation below: calculate the percent by mass of each element. %n = % %h = % %o = %
Answers: 3
You know the right answer?
If the number of moles of a gas initially contained in a 2.10 l vessel is doubled, what is the final...
Questions



Mathematics, 14.10.2019 20:30

Mathematics, 14.10.2019 20:30

History, 14.10.2019 20:30



English, 14.10.2019 20:30



History, 14.10.2019 20:30




History, 14.10.2019 20:30

Mathematics, 14.10.2019 20:30