
Chemistry, 21.07.2019 11:00 agarcia24101993
The voltage generated by the zinc concentration cell described by the line notation zn(s) ∣∣ zn2+(aq,0.100 m) ∥∥ zn2+(aq,? m) ∣∣ zn(s) is 14.0 mv at 25 °c. calculate the concentration of the zn2+(aq) ion at the cathode.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 16:00
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1

Chemistry, 22.06.2019 16:50
Ajet plane is speeding down the runway during takeoff. air resistance is not negligible. identify the forces on the jet.
Answers: 3

Chemistry, 22.06.2019 18:30
Which sample at stp has the same number of atoms as 18 liters of ne at stp
Answers: 1

Chemistry, 22.06.2019 20:00
What happens to the temperature of a substance when the average kinetic energy of its particles increases?
Answers: 3
You know the right answer?
The voltage generated by the zinc concentration cell described by the line notation zn(s) ∣∣ zn2+(aq...
Questions


Mathematics, 09.12.2020 17:10

Health, 09.12.2020 17:10


Mathematics, 09.12.2020 17:10

Law, 09.12.2020 17:10

Social Studies, 09.12.2020 17:10


Mathematics, 09.12.2020 17:10

Chemistry, 09.12.2020 17:10



Spanish, 09.12.2020 17:10

Mathematics, 09.12.2020 17:10



Geography, 09.12.2020 17:10

Chemistry, 09.12.2020 17:10

English, 09.12.2020 17:10

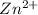 ion at cathode is 0.295 M
ion at cathode is 0.295 M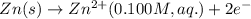
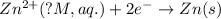
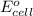 will be equal to zero.
will be equal to zero.![E_{cell}=E^o_{cell}-\frac{0.0592}{n}\log \frac{[Zn^{2+}]_{anode}}{[Zn^{2+}]_{cathode}}](/tpl/images/0115/3857/2e4c8.png)
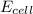 = 14.0 mV = 0.014 V (Conversion factor: 1 V = 1000 mV)
= 14.0 mV = 0.014 V (Conversion factor: 1 V = 1000 mV)![[Zn^{2+}]_{anode}](/tpl/images/0115/3857/a4cd7.png) = 0.100 M
= 0.100 M![[Zn^{2+}]_{cathode}](/tpl/images/0115/3857/54f88.png) = ? M
= ? M![0.014=0-\frac{0.0592}{2}\log \frac{0.100M}{[Zn^{2+}]_{cathode}}](/tpl/images/0115/3857/e07d0.png)
![[Zn^{2+}]_{cathode}=0.295M](/tpl/images/0115/3857/a683c.png)



