
Chemistry, 21.07.2019 15:00 Dmoney7784
Give the percent yield when 28.16 g of co2 are formed from the reaction of 4.000 moles of c8h18 with 4.000 moles of o2. 2 c8h18(l) + 25 o2(g) → 16 co2(g) + 18 h2o(g) molar mass co2 = 44.01 g/mol

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:40
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 13:00
How many moles of sulfur dioxide are produced when 4.38 moles of oxygen completely react with iron (iv) sulfide
Answers: 2

Chemistry, 22.06.2019 15:30
Which statement names the physical property of wood a. wood is softer than coal b. wood does not rust c. wood can rot d. wood can burn
Answers: 1

Chemistry, 23.06.2019 00:00
How many peaks will be present in a mass spectrum for brcl?
Answers: 1
You know the right answer?
Give the percent yield when 28.16 g of co2 are formed from the reaction of 4.000 moles of c8h18 with...
Questions

Computers and Technology, 17.06.2021 14:40

Business, 17.06.2021 14:40

Business, 17.06.2021 14:40

English, 17.06.2021 14:40







Engineering, 17.06.2021 14:40

Computers and Technology, 17.06.2021 14:40


Engineering, 17.06.2021 14:40


Mathematics, 17.06.2021 14:40

Mathematics, 17.06.2021 14:40

English, 17.06.2021 14:40


Geography, 17.06.2021 14:40

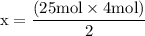 x = 50 moles of oxygen or O₂.
x = 50 moles of oxygen or O₂. 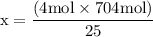 x = 112.64 gram of carbon dioxide
x = 112.64 gram of carbon dioxide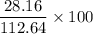 Percentage yield = 25%
Percentage yield = 25%

