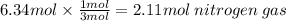Chemistry, 23.07.2019 07:30 valvaldeziv6373
The following balanced equation shows the formation of ammonia. n2 + 3h2 mc021-1.jpg 2nh3 how many moles of nitrogen are needed to completely convert 6.34 mol of hydrogen? 1.02 mol 2.11 mol 12.68 mol 19.02 mol

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:30
**40** points asapessay questions (10 points possible) clear image of next, create your own scenario. it can be one of your own real experiences or one you make up. use imagery in your writing to give your instructor a the setting and an action taking pace in your writing explain the structure and functions of the skin at work in your scenario. !
Answers: 3

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

Chemistry, 22.06.2019 05:30
Which other elements contain the same number of outer electrons as sodium
Answers: 3

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2
You know the right answer?
The following balanced equation shows the formation of ammonia. n2 + 3h2 mc021-1.jpg 2nh3 how many m...
Questions

Mathematics, 16.11.2020 19:40

Advanced Placement (AP), 16.11.2020 19:40

History, 16.11.2020 19:40



Mathematics, 16.11.2020 19:40


Mathematics, 16.11.2020 19:40




Social Studies, 16.11.2020 19:40


History, 16.11.2020 19:40


Mathematics, 16.11.2020 19:40

Chemistry, 16.11.2020 19:40

Mathematics, 16.11.2020 19:40

English, 16.11.2020 19:40

Physics, 16.11.2020 19:40