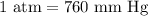Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
In the cathode ray tube experiment, j. j. thomson passed an electric current through different gases inside a cathode ray tube in the presence of an electric field. in which two ways did this experiment change scientists’ understanding of the atom?
Answers: 2

Chemistry, 22.06.2019 07:30
What is i fracture in the crust called when land move up, down or sideways
Answers: 2

Chemistry, 22.06.2019 17:10
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2

Chemistry, 23.06.2019 00:00
Mercury turns to a vapor at 629.88 k. how much heat is lost when 75.0 g of mercury vapor at 650 k condenses to a liquid at 297 k?
Answers: 1
You know the right answer?
What is the volume of a gas that exerts a pressure of 457 mm hg if it exerted a pressure of 2.50 atm...
Questions


Biology, 16.10.2019 03:00



Biology, 16.10.2019 03:00

Health, 16.10.2019 03:00

Mathematics, 16.10.2019 03:00


English, 16.10.2019 03:00

Biology, 16.10.2019 03:00


Social Studies, 16.10.2019 03:00

Physics, 16.10.2019 03:00



English, 16.10.2019 03:00

Biology, 16.10.2019 03:00



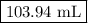 of gas exerts a pressure of 457 mm Hg.
of gas exerts a pressure of 457 mm Hg.
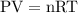 …… (1)
…… (1)
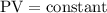 …… (2)
…… (2)
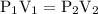 …… (3)
…… (3)
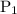 is the initial pressure.
is the initial pressure.
 is the final pressure.
is the final pressure.
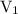 is the initial volume.
is the initial volume.
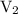 is the final volume.
is the final volume.
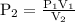 …… (4)
…… (4)
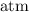 into
into 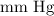 is as follows:
is as follows: