
Chemistry, 25.07.2019 07:30 ramentome7542
When a 3.80 g sample of magnesium nitride (mw 101g/mol) is reacted with 3.30 g of water, 3.60 g of mgo is formed. what is the percent yield of this reaction? mg3n2 + 3 h2o --> 2 nh3 + 3 mgo

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:00
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2

Chemistry, 22.06.2019 21:30
In one or two grammatically correct sentences, write a definition for the term molecule geometry
Answers: 3

Chemistry, 22.06.2019 23:00
If two identical atoms are bonded,what kind of molecule is formed
Answers: 1

Chemistry, 22.06.2019 23:00
Which of your 24 wells had indications that a chemical reaction occurred? how were you able to tell that a chemical reaction occurred? which of your 24 wells had indications that a physical reaction occurred? how were you able to tell that a physical reaction occurred? report on both mixing and evaporation. make a general statement about whether your hypotheses were validated or rejected. must your hypotheses be correct for this to be a successful laboratory?
Answers: 3
You know the right answer?
When a 3.80 g sample of magnesium nitride (mw 101g/mol) is reacted with 3.30 g of water, 3.60 g of m...
Questions

Mathematics, 15.02.2021 21:00



Mathematics, 15.02.2021 21:00



Mathematics, 15.02.2021 21:00

History, 15.02.2021 21:00

Mathematics, 15.02.2021 21:00


Mathematics, 15.02.2021 21:00


Social Studies, 15.02.2021 21:00

Mathematics, 15.02.2021 21:00


Mathematics, 15.02.2021 21:00



Mathematics, 15.02.2021 21:00

Chemistry, 15.02.2021 21:00

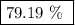 .
.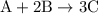
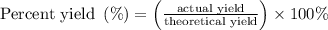 ......(1)
......(1)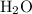 and is as follows:
and is as follows:
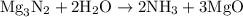 ……. (2)
……. (2)
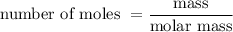
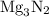 is
is 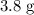 .
.
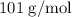 .
.
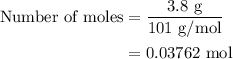
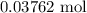 .
.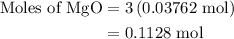
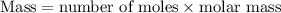
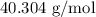 .
.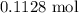 .
.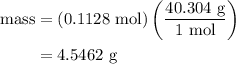
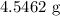 .
.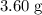 .
.
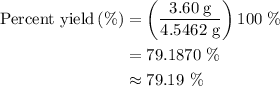
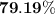 .
.


