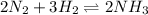Chemistry, 25.07.2019 13:30 pamelperezz26
For the reaction n2(g) + 3h2(g) 2nh3(g), what will happen if more ammonia gas is added? a.)there will be a shift toward the products. b.)the value for keq will change. c.)there will not be a shift in equilibrium. d.)there will be a shift toward the reactants.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:00
In any energy conversion, some of the energy is lost to the environment as question 5 options: electrical energy potential energy sound energy thermal energy
Answers: 1

Chemistry, 21.06.2019 21:30
If i make a solution by adding 83grams of sodium hydroxide to 750ml i’d water what is the molarity of sodium hydroxide
Answers: 1

Chemistry, 21.06.2019 22:30
Which statement best describes the oxidation numbers of the atoms found in magnesium chloride? a. magnesium has a 2- oxidation number and chlorine has a 1+ oxidation number. b. magnesium has a 2- oxidation number and chlorine has a 2+ oxidation number. c. magnesium has a 2+ oxidation number and chlorine has a 1- oxidation number. d. magnesium has a 1+ oxidation number and chlorine has a 1- oxidation number.
Answers: 2

Chemistry, 22.06.2019 01:30
In a spacecraft, the following reaction occurs: co2(g) + 2lioh(s) -> lico3(s) + h2o(i) (i attached picture of equation) how many liters of carbon dioxide will 4 moles of lithium hydroxide (lioh) absorb? (one mole of any gads occupies 22.4 l under certain conditions of temperature and pressure. assume those conditions for this equation.) 45l 6.0l 3.0l 34l
Answers: 1
You know the right answer?
For the reaction n2(g) + 3h2(g) 2nh3(g), what will happen if more ammonia gas is added? a.)there w...
Questions


History, 16.11.2019 23:31



Mathematics, 16.11.2019 23:31



Geography, 16.11.2019 23:31

History, 16.11.2019 23:31

Biology, 16.11.2019 23:31

Mathematics, 16.11.2019 23:31

Mathematics, 16.11.2019 23:31



Spanish, 16.11.2019 23:31

Mathematics, 16.11.2019 23:31

Mathematics, 16.11.2019 23:31



Mathematics, 16.11.2019 23:31