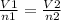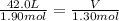Chemistry, 25.07.2019 14:00 brittany7436
Acylinder, with a piston pressing down with a constant pressure, is filled with 1.90 moles of a gas (n1), and its volume is 42.0 l (v1). if 0.600 mole of gas leak out, and the pressure and temperature remain the same, what is the final volume of the gas inside the cylinder?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 13:00
Jose and eric were given four samples in lab. the results of their analysis are shown in the table. based on the data they collected, which sample is most likely a metal?
Answers: 1

Chemistry, 23.06.2019 07:00
Under what conditions will a gas be most likely to exhibit the ideal gas properties predicted by the ideal gas law? 1)high pressures and high temperature, because particles are forced closer together with higher kinetic energy, so intermolecular forces of attraction are weaker 2)high pressure and low temperature, because particles are forced closer together and moving slower, so the volume of the particles is less significant 3) low pressure and high temperature, because particles are spread farther apart and moving faster, so the intermolecular forces of attraction are weaker 4)low pressure and low temperature, because particles are spread farther apart with lower kinetic energy, so the volume of the particles is less significant
Answers: 2

Chemistry, 23.06.2019 10:10
Solid tin exists in two forms: white and gray. for the transformation sn(s, white) → sn(s, gray) the enthalpy change is -2.1 kj/mol and the entropy change is -7.4 j/(mol*k). a. calculate the gibbs free energy change for the conversion of 1.00 mol white tin to gray tin at -30℃. b. will white tin convert spontaneously to gray tin at -30℃? c. at what temperature are white and gray tin thermodynamically equivalent at a pressure of 1 atm?
Answers: 3

Chemistry, 23.06.2019 10:30
Ethyl alcohol, also known as ethanol, has a density of 0.79 g/ml. what is the volume, in quarts, of 1.95 kg of this alcohol?
Answers: 2
You know the right answer?
Acylinder, with a piston pressing down with a constant pressure, is filled with 1.90 moles of a gas...
Questions


Biology, 17.04.2021 01:00



Social Studies, 17.04.2021 01:00

English, 17.04.2021 01:00


Social Studies, 17.04.2021 01:00

Geography, 17.04.2021 01:00


History, 17.04.2021 01:00

Mathematics, 17.04.2021 01:00


Mathematics, 17.04.2021 01:00


Physics, 17.04.2021 01:00

Mathematics, 17.04.2021 01:00



Mathematics, 17.04.2021 01:00