
Chemistry, 26.07.2019 13:30 jailenevazquez755
The solubility of agcl(s) in water at 25°c is 1.33 x 10-5 mol/l and its δh° of solution is 65.7 kj/mol. what is its solubility at 95°c?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:10
Explain the relationship between forward and backward reactions in equilibrium, and predict how changing the amount of a reactant (creating a tension) will affect that relationship.
Answers: 1

Chemistry, 22.06.2019 06:30
What is the correct term for living the most sustainable life you can within your current circumstances?
Answers: 1

Chemistry, 22.06.2019 07:00
The blackbody curve for a star name zeta is shown below. what is the peak wavelength for this star ?
Answers: 1

Chemistry, 22.06.2019 10:10
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate,m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3
You know the right answer?
The solubility of agcl(s) in water at 25°c is 1.33 x 10-5 mol/l and its δh° of solution is 65.7 kj/m...
Questions

Mathematics, 28.01.2021 23:40


French, 28.01.2021 23:40

Mathematics, 28.01.2021 23:40


Mathematics, 28.01.2021 23:40


SAT, 28.01.2021 23:40



Biology, 28.01.2021 23:40

Mathematics, 28.01.2021 23:40

Mathematics, 28.01.2021 23:40



Mathematics, 28.01.2021 23:40

French, 28.01.2021 23:40

Computers and Technology, 28.01.2021 23:40


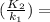 - ΔH∘ / R * ( 1 / T₂ - 1 / T₁ ) --- ( 1 )
- ΔH∘ / R * ( 1 / T₂ - 1 / T₁ ) --- ( 1 )


