Chemistry, 27.07.2019 09:30 brasherfamily14
What volume of a 1.0 m hcl is required to completely neutralize 25.0 ml of a 1.0m koh

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Write a brief passage describing a neutral atom of nitrogen-14 (n-14). describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. use the periodic table to you. 14 protons and eletrons since its a neutral atom
Answers: 1


Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1

Chemistry, 22.06.2019 13:00
Lab reagent, hypothesis test.a reference solution used as a lab reagent is purported to have a concentration of 5 mg/dl. six samples are taken from this solution and the following concentrations are recorded: (5.32, 4.88, 5.10, 4.73, 5.15, 4.75) mg/dl.these six measurements are assumed to be an srs of all possible measurements from solution.they are also assumed to have a standard deviation of 0.2, a normal distributin, and a mean concentration equal to the true concentration of the solution.carry out a significance test to determine whether these six measurements provide reliable evidence that the true concentration of the solution is actually not 5 mg/dl.
Answers: 1
You know the right answer?
What volume of a 1.0 m hcl is required to completely neutralize 25.0 ml of a 1.0m koh...
Questions


English, 30.08.2019 16:30


Health, 30.08.2019 16:30

Mathematics, 30.08.2019 16:30

Mathematics, 30.08.2019 16:30



Chemistry, 30.08.2019 16:30


Mathematics, 30.08.2019 16:30


Chemistry, 30.08.2019 16:30

Health, 30.08.2019 16:30

Business, 30.08.2019 16:30

Biology, 30.08.2019 16:30


Social Studies, 30.08.2019 16:30


Mathematics, 30.08.2019 16:30

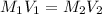
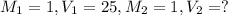
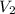 )
)