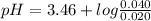Chemistry, 27.07.2019 19:30 brittanyfox411
Calculate the ph of a buffer that is 0.020 m hf and 0.040 m naf. the ka for hf is 3.5 × 10-4.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:10
Using complete sentences, explain how to predict the products and balance the reaction between sulfuric acid and potassium hydroxide.
Answers: 1



Chemistry, 22.06.2019 22:20
Asuspension of yeast cells is being grown under anaerobic conditions such that glucose is degraded to ethanol and carbon dioxide. if one wishes to follow this process by monitoring the release of 14co2, at which positions in the glucose molecule would the 14c label need to be incorporated?
Answers: 2
You know the right answer?
Calculate the ph of a buffer that is 0.020 m hf and 0.040 m naf. the ka for hf is 3.5 × 10-4....
Questions

Mathematics, 18.03.2021 03:10

Mathematics, 18.03.2021 03:10


Mathematics, 18.03.2021 03:10



Mathematics, 18.03.2021 03:10





History, 18.03.2021 03:10


Mathematics, 18.03.2021 03:10






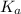 of HF is
of HF is 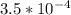
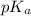 :
: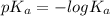
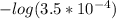
![pH = pK_{a} + log\frac{[Base]}{[Acid]}](/tpl/images/0139/8795/8fa71.png)