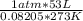Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:10
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3

Chemistry, 22.06.2019 05:30
Liv sheldon given the balanced equation for an organic reaction: c2h2 + 2cl2 → c2h2cl4 this reaction is best classified as *
Answers: 1

Chemistry, 22.06.2019 12:30
If 22.5 liters of oxygen reacted with excess of hydrogen, how many liters of water vapor could be produced?
Answers: 3

Chemistry, 22.06.2019 20:00
How are the terms group and period used on the periodic table
Answers: 1
You know the right answer?
How many moles of o2 are in 53 l of o2 at stp? a) 2.4 b) 0.45 c) 1.65 d) 1187...
Questions


History, 05.02.2021 20:30





Mathematics, 05.02.2021 20:30


Mathematics, 05.02.2021 20:30


Mathematics, 05.02.2021 20:30


History, 05.02.2021 20:30




Mathematics, 05.02.2021 20:30


History, 05.02.2021 20:30

Mathematics, 05.02.2021 20:30

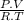 =
=