Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2

Chemistry, 23.06.2019 08:00
What is the temperature in kelvin of a gas if it is allowed to expand from 1.50 l to 4.50 l? the initial temperature is 10.0°c and pressure is constant throughout the change. which equation should you use? t2= v2/v1 t1 what is the final temperature? ⇒ 849 k these are the answers.
Answers: 1
You know the right answer?
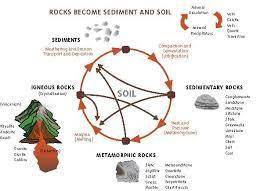
The formation of soil is primarily the result of...
Questions

Computers and Technology, 08.12.2021 20:40



SAT, 08.12.2021 20:40



History, 08.12.2021 20:40


Computers and Technology, 08.12.2021 20:40


Social Studies, 08.12.2021 20:40


Law, 08.12.2021 20:40

Arts, 08.12.2021 20:40


Mathematics, 08.12.2021 20:40

Mathematics, 08.12.2021 20:40

English, 08.12.2021 20:40