Chemistry, 29.07.2019 12:30 sipstick9411
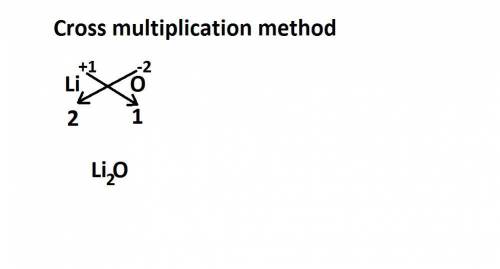
Lithium is a metal with an oxidation number of 1+ and oxygen is a nonmetal with an oxidation number of 2-. which chemical formula represents the compound formed by these two elements? a. li2o b. li3o c. lio d. lio2

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:30
Balance this equation co2(g) + h2o (g) show that the balanced equation obeys the law if conversation of mass
Answers: 1

Chemistry, 21.06.2019 21:30
How air particles exert a pressure on the inside of the balloon
Answers: 1

Chemistry, 22.06.2019 03:30
If a solution is considered basic, then a) the hydroxide ion and hydronium ion concentrations are equal. b) the hydroxide ion concentration is less than the hydronium ion concentration. c) the hydronium ion concentration is greater than the hydroxide ion concentration. d) the hydroxide ion concentration is greater than the hydronium ion concentration.
Answers: 1

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3
You know the right answer?
Lithium is a metal with an oxidation number of 1+ and oxygen is a nonmetal with an oxidation number...
Questions


History, 21.07.2019 16:00

English, 21.07.2019 16:00


Mathematics, 21.07.2019 16:00

English, 21.07.2019 16:00

Spanish, 21.07.2019 16:00




Chemistry, 21.07.2019 16:00

Social Studies, 21.07.2019 16:00

Mathematics, 21.07.2019 16:00


Mathematics, 21.07.2019 16:00



History, 21.07.2019 16:00

Mathematics, 21.07.2019 16:00

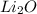 .
.