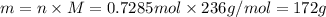Chemistry, 29.07.2019 14:00 bbenaventbbbb9653
Determine the mass of al(c2h3o2)3 that contains 2.63 χ 1024 atoms of oxygen.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Anurse practitioner prepares an injection of promethazine, an antihistamine used to treat allergic rhinitis. if the stock bottle is labeled 25 mg/ml and the order is a dose of 11.0 mg , how many milliliters will the nurse draw up in the syringe?
Answers: 3

Chemistry, 21.06.2019 18:30
For each of the following mixtures decide if filtering would be suitable to separate the substances. explain your answers. oil in water sugar in water sand in water chalk in water tea leaves in a cup of tea
Answers: 2

Chemistry, 21.06.2019 23:00
City a and city b had two different temperatures on a particular day. on that day, four times the temperature of city a was 8â° c more than 3 times the temperature of city b. the temperature of city a minus twice the temperature of city b was â’3â° c. what was the temperature of city a and city b on that day? city a was 5â° c, and city b was 4â° c. city a was 3â° c, and city b was â’1â° c. city a was 8â° c, and city b was â’3â° c. city a was 5â° c, and city b was â’5â° c.
Answers: 2

You know the right answer?
Determine the mass of al(c2h3o2)3 that contains 2.63 χ 1024 atoms of oxygen....
Questions

Mathematics, 17.12.2019 08:31

History, 17.12.2019 08:31

Mathematics, 17.12.2019 08:31



Mathematics, 17.12.2019 08:31




History, 17.12.2019 08:31





World Languages, 17.12.2019 08:31



Mathematics, 17.12.2019 08:31

History, 17.12.2019 08:31

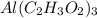 .
. 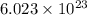 atoms of
atoms of 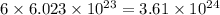 atoms of oxygen atoms.
atoms of oxygen atoms. 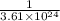 moles of
moles of 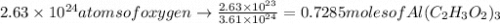 .
.