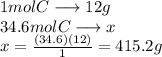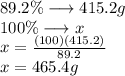Chemistry, 31.07.2019 22:00 Wolfgirl2032
Asubstance is 89.2% carbon by mass. how much of the substance would be needed to recover 34.6 mol of pure carbon?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Determine the wavelength of the light absorbed when an electron in a hydrogen atom makes a transition from an orbital in the n=3 level to an orbital in the n=7 level.
Answers: 2

Chemistry, 22.06.2019 13:30
1) which of the following is the best example of a physical change? a) sugar dissolving in tea b) firefly glowing 2) in the combustion of ethane, what is/are the reactants? c2h6 + o2 ==> co2 + h2o a) c2h6 and o2 b) co2 and c2h6
Answers: 2

Chemistry, 22.06.2019 18:20
Which reason best explains why metals are malleable? a)because they have delocalized electrons b)because they have localized electrons c)because they have ionic bonds d)because they have rigid bonds
Answers: 2

Chemistry, 22.06.2019 20:30
Some familiar products contain some of the same types of atoms. for instance, the chemical formula for baking soda is nahco 3. the chemical formula for liquid bleach is naclo, and the chemical formula for table salt is nacl. which choice best describes why these three products have some of the same types of atoms in common?
Answers: 1
You know the right answer?
Asubstance is 89.2% carbon by mass. how much of the substance would be needed to recover 34.6 mol of...
Questions

Mathematics, 06.11.2019 13:31

World Languages, 06.11.2019 13:31


Mathematics, 06.11.2019 13:31

English, 06.11.2019 13:31

Health, 06.11.2019 13:31




Biology, 06.11.2019 13:31





Business, 06.11.2019 13:31


Social Studies, 06.11.2019 13:31


Physics, 06.11.2019 13:31