4na + o2 > 2 na2o
for the reaction whose balanced equation is as follows, find the number...

Chemistry, 31.01.2020 10:56 Foxfire5109
4na + o2 > 2 na2o
for the reaction whose balanced equation is as follows, find the number of grams of sodium that must react to produce 42.0 grams of sodium oxide.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:30
For the following dehydrohalogenation (e2) reaction, draw the zaitsev product(s) resulting from elimination involving c3–c4 (i.e., the carbon atoms depicted with stereobonds). show the product stereochemistry clearly. if there is more than one organic product, both products may be drawn in the same box. ignore elimination involving c3 or c4 and any carbon atom other than c4 or c3.
Answers: 3

Chemistry, 22.06.2019 04:30
Which statement best describes the relationship between period and frequency of light waves? a) in wave b the period increases and the frequency decreases from wave a. b) in wave a the period increases and the frequency decreases from wave b. c) in wave b the period is shorter and the frequency is greater than in wave a. d) in wave a the period is shorter and the frequency is greater than in wave b.
Answers: 1

Chemistry, 22.06.2019 05:00
Agas can holds 2.0 gal of gasoline. what is this quantity in cubic centimeters?
Answers: 2

Chemistry, 22.06.2019 13:10
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1
You know the right answer?
Questions


Mathematics, 13.01.2021 19:50






Chemistry, 13.01.2021 19:50

Mathematics, 13.01.2021 19:50



History, 13.01.2021 19:50



Mathematics, 13.01.2021 19:50


Mathematics, 13.01.2021 19:50

Mathematics, 13.01.2021 19:50



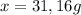 of Na
of Na

