
Chemistry, 03.08.2019 04:00 jjaheimhicks3419
What element forms an ion with an electronic configuration of 1s22s22p63s23p63d104s24p64d105s25p6 (or [xe]) and a –2 charge?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 12:30
Asap! ! who wants to me with my assignment : ) (especially with how i’m gonna draw out the example) you have explored some interesting, informative, and amusing examples of models. now it's time to get creative and make your own model. here is the requirement checklist for your model: ✔ model types can include drawings, diagrams, physical models, virtual simulations, or videos. ✔ models must be created by you, not something selected from an online or outside source. ✔ submit a presentation, picture, video, or screenshot of your model. ✔ submit a one-paragraph summary describing the topic you chose, your model, what it represents, how you made it, and the specific science involved. it is important that you are using science terminology and are accurate. now that you know how to create and submit your model, you will need to choose a topic for your model. choose one of the three topics listed below. select each topic for an overview. -conservation of mass -atomic theory -thermal energy
Answers: 2

Chemistry, 22.06.2019 06:30
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1

Chemistry, 22.06.2019 13:10
What type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? view available hint(s) what type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? salt bridge disulfide bridge hydrogen bond hydrophobic interaction
Answers: 1

Chemistry, 22.06.2019 20:30
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3
You know the right answer?
What element forms an ion with an electronic configuration of 1s22s22p63s23p63d104s24p64d105s25p6 (o...
Questions

Business, 17.09.2019 16:00




Social Studies, 17.09.2019 16:00





Mathematics, 17.09.2019 16:00

Business, 17.09.2019 16:00

English, 17.09.2019 16:00


Biology, 17.09.2019 16:00

English, 17.09.2019 16:00

Mathematics, 17.09.2019 16:00

Mathematics, 17.09.2019 16:00


Mathematics, 17.09.2019 16:00

Biology, 17.09.2019 16:00

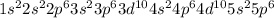 .
. 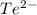 is as follows.
is as follows. 

