Chemistry, 04.08.2019 00:30 bullockarwen
Magnesium burns in air with a dazzling brilliance to produce magnesium oxide: 2mg(s) + o2(g) → 2mgo(s) how many moles of o2 are consumed when 0.550 mol of magnesium burns?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Particles vibrate in a rigid structure and do not move relative to their neighbors.
Answers: 1

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 2

Chemistry, 22.06.2019 23:00
What is the oxidation state of each individual carbon atom in c2o42−?
Answers: 1

Chemistry, 23.06.2019 07:30
In the diagram here that represents the reaction, which reactant, a or b, is the limiting reagent?
Answers: 1
You know the right answer?
Magnesium burns in air with a dazzling brilliance to produce magnesium oxide: 2mg(s) + o2(g) → 2mgo...
Questions









Advanced Placement (AP), 03.12.2019 23:31



Mathematics, 03.12.2019 23:31


Mathematics, 03.12.2019 23:31






Medicine, 03.12.2019 23:31

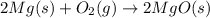
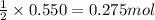 of oxygen gas.
of oxygen gas.