
Chemistry, 04.08.2019 16:00 NeonPlaySword
One of the reactions for rusting iron is as follows: 4fe + 3o2 → 2fe2o3 (mm fe: 55.85 g/mol; mm o2=32 g/mol; mm fe2o3=159.70 g/mol) if 63.98 g of oxygen gas is completely consumed, how many moles of iron (iii) oxide are formed? a. 1.333 mol b. 3071 mol c. 2.999 mol d. 6812 mol

Answers: 1


Another question on Chemistry




Chemistry, 22.06.2019 19:30
To calculate percent by mass, use the equation below: calculate the percent by mass of each element. %n = % %h = % %o = %
Answers: 3
You know the right answer?
One of the reactions for rusting iron is as follows: 4fe + 3o2 → 2fe2o3 (mm fe: 55.85 g/mol; mm...
Questions


World Languages, 09.12.2020 22:10

Mathematics, 09.12.2020 22:10

Mathematics, 09.12.2020 22:10


History, 09.12.2020 22:10

Mathematics, 09.12.2020 22:10







Mathematics, 09.12.2020 22:10

Biology, 09.12.2020 22:10



History, 09.12.2020 22:10

Mathematics, 09.12.2020 22:10

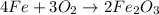
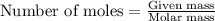
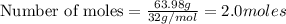
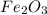 .
.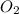 produces 2 moles of
produces 2 moles of 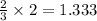 moles of
moles of 

