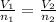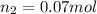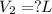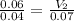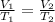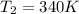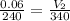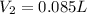Chemistry, 04.08.2019 06:50 dchirunga23
For this exercise, you can simulate the described conditions by changing the values in the run experiment tool of the simulation. to be able to measure the effects on the gas volume, slide the rspd bar in the properties box to v (l) so that the volume bar turns yellow. this way, volume becomes the only dependent variable. note that when volume is selected with rspd, its value can no longer be directly controlled. suppose a piston automatically adjusts to maintain a gas at a constant pressure of 14.20 atm . for the initial conditions, there are 0.04 mol of helium at a temperature of 240.00 k . this gas occupies a volume of 0.06 l under those conditions. what volume will the gas occupy if the number of moles is increased to 0.07 mol (n2) from the initial conditions? what volume will the gas occupy if the temperature is increased to 340.00 k (t2) from the initial conditions?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:00
Agas in a balloon at constant pressure has a volume of 160 ml at -125*c. what is its volume at 29.0*c?
Answers: 1

Chemistry, 22.06.2019 12:00
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1

Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1

Chemistry, 22.06.2019 23:10
Afusion reaction takes place between carbon and another element. neutrons are released, and a different element is formed. the different element is a) lighter than helium.b)heavier than helium.c)the same weight as helium.d)dependent on the element that reacted with carbon.
Answers: 3
You know the right answer?
For this exercise, you can simulate the described conditions by changing the values in the run exper...
Questions


Mathematics, 30.01.2020 21:46


Arts, 30.01.2020 21:46



History, 30.01.2020 21:46





Mathematics, 30.01.2020 21:46

History, 30.01.2020 21:47




Mathematics, 30.01.2020 21:47

Physics, 30.01.2020 21:47


Chemistry, 30.01.2020 21:47