Chemistry, 02.08.2019 05:30 isabella4141
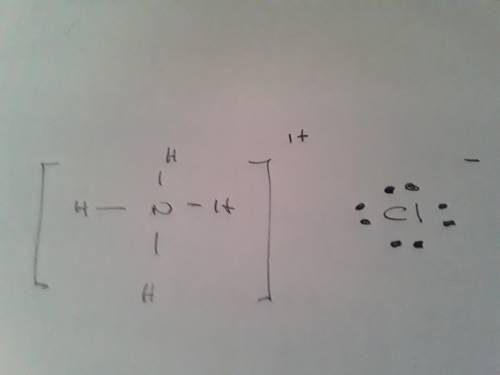
Ammonium chloride, nh4cl, is a very soluble salt in water. draw the lewis structures of the ammonium and chloride ions. draw the molecule by placing atoms on the grid and connecting them with bonds. include all lone pairs of electrons. show the formal charges of all nonhydrogen atoms in the correct structure.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:40
1) in saturated limewater, [h+ ]=3.98x10-13 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 2) in butter, [h+ ]=6.0x10-7 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 3) in peaches, [oh]=3.16x10-11 m a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 4) during the course of the day, human saliva varies between being acidic and basic. if [oh]=3.16x10-8 m, a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? /
Answers: 3

Chemistry, 23.06.2019 07:30
The compound formed from 2 atoms of hydrogen and one atom of oxygen is
Answers: 1

Chemistry, 23.06.2019 12:30
What would happen to a weak base dissociation equilibrium if more products we added
Answers: 1

Chemistry, 23.06.2019 21:30
Acertain substance has a solubility of 12 grams in 100 grams of water at 20°c. this means that the substance will begin to dissolve when 12 grams are present in solution when 12 grams of the substance are stirred into a beaker with 100 g of water, it will begin settling at the bottom when 12 g of the substance are dissolved in 100 grams of water, the solution will be dilute when 12 g of the substance are dissolved in 100 grams of water, the solution will be saturated
Answers: 1
You know the right answer?
Ammonium chloride, nh4cl, is a very soluble salt in water. draw the lewis structures of the ammonium...
Questions










Mathematics, 05.11.2020 21:10


Mathematics, 05.11.2020 21:10

Computers and Technology, 05.11.2020 21:10

History, 05.11.2020 21:10



Mathematics, 05.11.2020 21:10

History, 05.11.2020 21:10

Chemistry, 05.11.2020 21:10