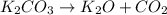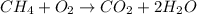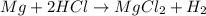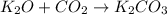Chemistry, 01.08.2019 07:10 alangonsales6232
Which of the following statements about decomposition reactions is true? a) decomposition and combustion reactions are the same thing b) decomposition and single-displacement reactions are the same thing c) decomposition reactions are essentially the opposite of synthesis reactions d) decomposition reactions are essentially the opposite of single-displacement reactions

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:00
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1

Chemistry, 21.06.2019 23:00
What is the maximum amount of al2(so4)3 which could be formed from 15.84 g of al and 12.89 g of cuso4?
Answers: 2

Chemistry, 22.06.2019 12:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al oxidizing agent = reducing agent =
Answers: 1

Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2
You know the right answer?
Which of the following statements about decomposition reactions is true? a) decomposition and combu...
Questions




Mathematics, 26.04.2021 22:40


Mathematics, 26.04.2021 22:40






History, 26.04.2021 22:40



Mathematics, 26.04.2021 22:40


Mathematics, 26.04.2021 22:40