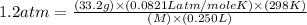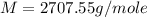Chemistry, 27.07.2019 18:40 krazziekidd2p845ri
An aqueous solution of a soluble compound (a nonelectrolyte) is prepared by dissolving 33.2 g of the compound in sufficient water to form 250 ml of solution. the solution has an osmotic pressure of 1.2 atm at 25 °c. what is the molar mass (g/mole) of the compound?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:30
Calculate the molar mass of aluminum oxide (al2o3). express your answer to four significant figures.
Answers: 1

Chemistry, 22.06.2019 14:00
Calculate the frequency of a wave in a spring toy. the wave has a speed of 1.1 meters per second and a wavelength of 0.1 meters. *
Answers: 2

Chemistry, 22.06.2019 15:30
Why does earth rotate? because earth is formed from cold gases collapsing due to gravity because the matter in the nebula that formed earth was spinning because earth forms more than 99% of the mass of the solar system because the hydrogen atoms inside the nebula fused to form helium
Answers: 1

Chemistry, 23.06.2019 01:00
Which statement best describes isomers? a. isomers are alcohols that have the same functional group. b. isomers have at least one carbon-carbon double bond. c. isomers have the same molecular formula but different structural properties.
Answers: 1
You know the right answer?
An aqueous solution of a soluble compound (a nonelectrolyte) is prepared by dissolving 33.2 g of the...
Questions

Chemistry, 10.06.2021 01:00


Biology, 10.06.2021 01:00

Biology, 10.06.2021 01:00

English, 10.06.2021 01:00

Mathematics, 10.06.2021 01:00


Mathematics, 10.06.2021 01:00

Mathematics, 10.06.2021 01:00

English, 10.06.2021 01:00

Social Studies, 10.06.2021 01:00

Mathematics, 10.06.2021 01:00



Mathematics, 10.06.2021 01:00

Mathematics, 10.06.2021 01:00

Mathematics, 10.06.2021 01:00

Mathematics, 10.06.2021 01:00

Mathematics, 10.06.2021 01:00

Chemistry, 10.06.2021 01:00

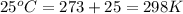
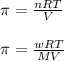
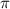 = osmotic pressure
= osmotic pressure