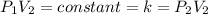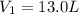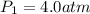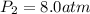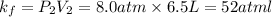Chemistry, 23.07.2019 09:40 catalanb474
Acertain gas is present in a 13.0 l cylinder at 4.0 atm pressure. if the pressure is increased to 8.0 atm , the volume of the gas decreases to 6.5 l . find the two constants ki, the initial value of k, and kf, the final value of k, to verify whether the gas obeys boyle's law. express your answers to two significant figures separated by a comma.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which traits do human embryos have that link them to a common ancestor with fish and reptiles? a. scales and tail b. gill slits and scales c. tail and gill slits d. hair and tail
Answers: 2

Chemistry, 22.06.2019 16:30
At 20°c, a sample of h2o liquid and a sample of co2 gas each have the same average kinetic energy. why is one a liquid and the other a gas at this temperature?
Answers: 1


Chemistry, 22.06.2019 23:00
What is the oxidation state of an individual bromine atom in nabro3?
Answers: 2
You know the right answer?
Acertain gas is present in a 13.0 l cylinder at 4.0 atm pressure. if the pressure is increased to 8....
Questions


Social Studies, 31.07.2019 10:10



Mathematics, 31.07.2019 10:10

Biology, 31.07.2019 10:10






Business, 31.07.2019 10:10



Biology, 31.07.2019 10:10





Geography, 31.07.2019 10:20

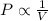 (At constant temperature and number of moles)
(At constant temperature and number of moles)