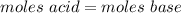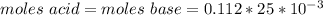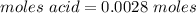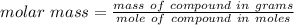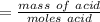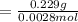Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:30
The melting point of sulfur is 115 °c and its boiling point is 445 °c. what state would sulfur be in at 200 °c?
Answers: 1

Chemistry, 22.06.2019 14:00
Ascientist measures the speed of sound in a monatomic gas to be 449 m/s at 20∘c. what is the molar mass of this gas?
Answers: 2

Chemistry, 22.06.2019 19:30
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2

You know the right answer?
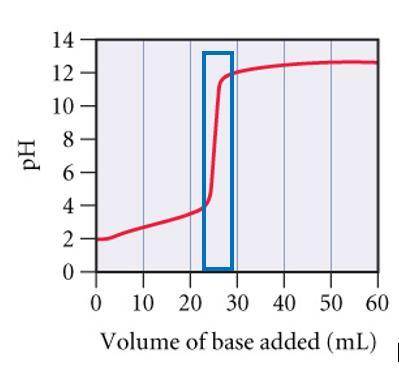
A0.229-g sample of an unknown monoprotic acid is titrated with 0.112 m naoh. the resulting titration...
Questions


Social Studies, 29.08.2019 02:30

Biology, 29.08.2019 02:30

History, 29.08.2019 02:30

Mathematics, 29.08.2019 02:30

Mathematics, 29.08.2019 02:30


Mathematics, 29.08.2019 02:30

Mathematics, 29.08.2019 02:30


Biology, 29.08.2019 02:30

Geography, 29.08.2019 02:30

Physics, 29.08.2019 02:30


English, 29.08.2019 02:30

Mathematics, 29.08.2019 02:30

Mathematics, 29.08.2019 02:30


Chemistry, 29.08.2019 02:30

Biology, 29.08.2019 02:30