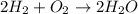Chemistry, 15.07.2019 17:40 brooklyn674
Read the scenario. two molecules of hydrogen gas and one molecule of oxygen gas react to yield two molecules of water. which of the following equations properly models the reaction described? h2 + o2 → h2o2 h2 + o2 → 2h2o h + o → h2o 2h2 + o2 → 2h2o

Answers: 1


Another question on Chemistry



Chemistry, 23.06.2019 02:20
In a chemical reaction, the final amount of the products is determined by the a. universal gas law b. law of definite proportions c. air pressure d. temperature e. none of the above me
Answers: 2

Chemistry, 23.06.2019 04:00
If you are told to get 100 ml of stock solution to use to prepare smaller size sample for an experiment, which piece of glassware would you use?
Answers: 1
You know the right answer?
Read the scenario. two molecules of hydrogen gas and one molecule of oxygen gas react to yield two...
Questions

Mathematics, 28.08.2019 17:30

Mathematics, 28.08.2019 17:30

Physics, 28.08.2019 17:30

English, 28.08.2019 17:30


History, 28.08.2019 17:30



Mathematics, 28.08.2019 17:30

Mathematics, 28.08.2019 17:30





History, 28.08.2019 17:30

Mathematics, 28.08.2019 17:30


Mathematics, 28.08.2019 17:30