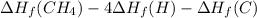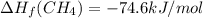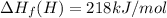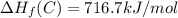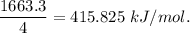Chemistry, 11.07.2019 05:50 jodonw1955
Calculate the average molar bond enthalpy of the carbon-hydrogen bond in a ch4 molecule.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 22.06.2019 08:00
Identify a strong intermolecular force of attraction between an alcohol
Answers: 1

Chemistry, 22.06.2019 15:30
Count the number of each type of atom in the equation below, and then balance the equation. write in the numbers of atoms and coefficients. add a 1 if there should be no coefficient. cs2(l) + o2(g) → co2(g) + so2(g) c [ ] s [ ] o > c [ ] s [ ] o [ ] cs2(l) + [ ] o2(g) > [ ] co2(g) + [ ] so2(g)
Answers: 3

Chemistry, 22.06.2019 17:00
According to the kinetic-molecular theory, what happens to a liquid when it is transferred from one container to another? the volume and the shape stay the same. the volume increases to fill the new container, but the shape stays the same. the volume stays the same, but the shape changes to fit the new container. the volume and the shape change to fill the new container.
Answers: 2
You know the right answer?
Calculate the average molar bond enthalpy of the carbon-hydrogen bond in a ch4 molecule....
Questions


Mathematics, 01.07.2019 05:20

Mathematics, 01.07.2019 05:20

Mathematics, 01.07.2019 05:20

Mathematics, 01.07.2019 05:20


History, 01.07.2019 05:20

Mathematics, 01.07.2019 05:20


Arts, 01.07.2019 05:20

Mathematics, 01.07.2019 05:20

Biology, 01.07.2019 05:20

Mathematics, 01.07.2019 05:20

Mathematics, 01.07.2019 05:20



History, 01.07.2019 05:20

Mathematics, 01.07.2019 05:20

History, 01.07.2019 05:20

Mathematics, 01.07.2019 05:20