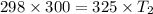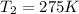Chemistry, 05.07.2019 09:00 natalie2sheffield
The pressure in a car tire is 298 kilopascals at 300 kelvin. after a long drive, the pressure becomes 325 kilopascals. which law will you use to calculate the new temperature in the tire assuming that the volume is constant?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 10:10
How do you identify the anode on a power source such as a battery? how do you identify the cathode? how are terms anion and cation?
Answers: 1


Chemistry, 22.06.2019 14:30
Which of the following describes a situation where competition between producers exists
Answers: 1
You know the right answer?
The pressure in a car tire is 298 kilopascals at 300 kelvin. after a long drive, the pressure become...
Questions


Mathematics, 19.08.2020 20:01


History, 19.08.2020 20:01







Mathematics, 19.08.2020 20:01




Mathematics, 19.08.2020 20:01





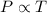 (At constant volume and number of moles)
(At constant volume and number of moles)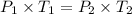
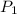 = initial pressure of gas = 298 kPa
= initial pressure of gas = 298 kPa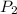 = final pressure of gas = 325 kPa
= final pressure of gas = 325 kPa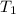 = initial temperature of gas = 300 K
= initial temperature of gas = 300 K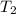 = final temperature of gas = ?
= final temperature of gas = ?