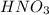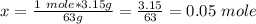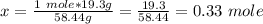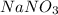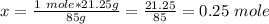Chemistry, 18.08.2019 19:30 tinyiaihfurlow
Calculate the number of moles 3.15 g of hno3 19.3 g of nacl 21.25 g of nano3

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:10
3.) for each of the following compounds, draw the major organic product of reaction with hcl or naoh and circle whether the starting materials and products will be more soluble in organic solvent or water benzoic acid + hcl: benzoic acid + naoh: oh benzoic acid water/organic water organic fluorenone hс: fluorenone + naoh: fluorenone water/organic water/organic веnzocaine + hci: benzocaine + n»oh: h2n benzocaine water/organic water organic o=
Answers: 3

Chemistry, 22.06.2019 07:30
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3


Chemistry, 22.06.2019 09:30
Apump contains 0.5 l of air at 203 kpa.you draw back on the piston of the pump, expanding the volume until the pressure reads 50.8 kpa. what is the new volume of the air pump
Answers: 2
You know the right answer?
Calculate the number of moles 3.15 g of hno3 19.3 g of nacl 21.25 g of nano3...
Questions


Mathematics, 25.05.2021 17:20


Mathematics, 25.05.2021 17:20

Mathematics, 25.05.2021 17:20

History, 25.05.2021 17:20

Mathematics, 25.05.2021 17:20




History, 25.05.2021 17:20


Mathematics, 25.05.2021 17:20

Mathematics, 25.05.2021 17:20

English, 25.05.2021 17:20


Mathematics, 25.05.2021 17:20