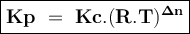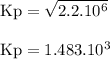Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:10
According to the diagram; a) identify the anode of the cell and write the half-reaction that occurs there b) write the overall equation for the reaction that occurs as the cell operates c) calculate the value of the standard cell potential ,e cell. d)write the shorthand notation of the cell above e)indicate the flow of the electrons on the diagram
Answers: 3


Chemistry, 22.06.2019 19:50
If a gas has an initial pressure of 101kpa and a volume of 10l, then it expands to a volume of 20l, what is the new pressure?
Answers: 2

Chemistry, 23.06.2019 06:00
•what conclusions can you make about the relationship between the volume of a gas and its temperature? • what conclusions can you make about the relationship between the volume of a gas and its pressure? • what possible variables have you not accounted for? as you did the procedures, is it possible that the atmospheric pressure may have changed? if it did change over the course of your experiment, then how would your results have been affected?
Answers: 3
You know the right answer?
The reaction below has an equilibrium constant kp=2.2×106 at 298 k. 2cof2(g)⇌co2(g)+cf4(g) calculate...
Questions



Mathematics, 15.10.2019 11:30

Mathematics, 15.10.2019 11:30

Chemistry, 15.10.2019 11:30


Physics, 15.10.2019 11:30

Chemistry, 15.10.2019 11:30



Business, 15.10.2019 11:30

Social Studies, 15.10.2019 11:30



Mathematics, 15.10.2019 11:30

Mathematics, 15.10.2019 11:30

Physics, 15.10.2019 11:30

Mathematics, 15.10.2019 11:30

English, 15.10.2019 11:30

![\large {\boxed {\bold {Kc ~ = ~ \frac {[C] ^ m [D] ^ n} {[A] ^ p [B] ^ q}}}}](/tpl/images/0052/1661/9d1db.png)
![\large {\boxed {\bold {Kp ~ = ~ \frac {[pC] ^ m [pD] ^ n} {[pA] ^ p [pB] ^ q}}}}](/tpl/images/0052/1661/b3cf6.png)