
Chemistry, 02.07.2019 14:50 connorhoran05
Calculate the resulting ph if 365 ml of 2.88 m hno3 is mixed with 335 ml of 1.10 m ca(oh)2 solution. be aware of stoichiometry!

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Write two balanced equations 1. dissolving of solid sodium hydroxide in water 2. the reaction of sodium hydroxide solution with hydrochloric acid
Answers: 1

Chemistry, 22.06.2019 02:00
If you add 10ml of hot water to 10ml of cold water and the change in tempature 8°c calculate how much energy is gained by the cold water
Answers: 1

Chemistry, 22.06.2019 11:00
As air becomes more dense, (select all that apply) o. air weighs less o. gas molecules are closer together o. air is colder o. air weighs more o. gas molecules are further apart o. air is hotter
Answers: 3

Chemistry, 22.06.2019 12:20
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3
You know the right answer?
Calculate the resulting ph if 365 ml of 2.88 m hno3 is mixed with 335 ml of 1.10 m ca(oh)2 solution....
Questions


Mathematics, 29.07.2019 05:00


French, 29.07.2019 05:00

Mathematics, 29.07.2019 05:00

Mathematics, 29.07.2019 05:00



Social Studies, 29.07.2019 05:00



History, 29.07.2019 05:00








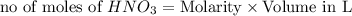
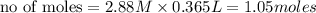
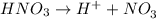
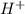 ions = 1.05
ions = 1.05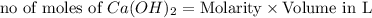
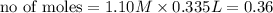
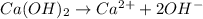
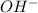 ions =
ions = 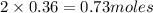
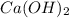 will neutralize 0.73 moles of
will neutralize 0.73 moles of 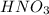 and (1.05-0.73)=0.32 moles of
and (1.05-0.73)=0.32 moles of 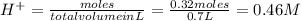
![pH=-\log[H^+]=-\log[0.46]=0.34](/tpl/images/0042/8951/36f67.png)


