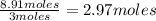Answers: 2


Another question on Chemistry


Chemistry, 23.06.2019 10:20
Based on the equation, how many grams of br2 are required to react completely with 29.2 grams of alcl3? alcl3 + br2 → albr3 + cl2 48.7 grams 52.6 grams 56.7 grams 61.3 grams
Answers: 3

Chemistry, 23.06.2019 10:30
Ireally need ! calcium metal reacts with a potassium chloride solution to form calcium chloride and potassium ions. balance this reaction. (s) + (aq) → cacl2(s) + +(aq) a) 1, 2, 1, 2 b) 1, 2, 1, 1 c) 1, 1, 1, 1 d) 2, 1, 2, 1
Answers: 1

Chemistry, 23.06.2019 17:00
Using this reversible reaction, answer the questions below: n2o4 2no2 (colorless) (reddish-brown) -as the temperature increased, what happened to the n2o4 concentration? -was the formation of reactants or products favored by the addition of heat? -which reaction is exothermic? right to left or left to right? -if the change of enthalpy of this reaction when proceeding left to right is 14 kcal, which chemical equation is correct? n2o4 2no2 + 14 kcal n2o4 2no2, hr = +14 kcal n2o4 + 14 kcal 2no2 n2o4 2no2, hr = -14 kcal
Answers: 1
You know the right answer?
Achemist needs to find out how large of a container would be required to contain the products of an...
Questions


Mathematics, 26.01.2020 23:31





Mathematics, 26.01.2020 23:31

History, 26.01.2020 23:31

Biology, 26.01.2020 23:31

Mathematics, 26.01.2020 23:31




Mathematics, 26.01.2020 23:31


Mathematics, 26.01.2020 23:31




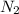 produced are, 2.97 moles
produced are, 2.97 moles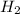 = 8.91 moles
= 8.91 moles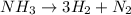
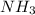 decomposes to produce 3 moles of
decomposes to produce 3 moles of