
Chemistry, 01.07.2019 14:00 conyabrew82
Which substance will have the largest temperature change if the same amount of heat is added to each of them? ethanol, c2h5oh(l) water, h2o(l) aluminum, al(s) iron, fe(s)?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 09:40
Which diagram shows the correct way to represent an ionic compound of magnesium oxide?
Answers: 3

Chemistry, 22.06.2019 10:10
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate,m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3
You know the right answer?
Which substance will have the largest temperature change if the same amount of heat is added to each...
Questions


Geography, 03.09.2020 21:01

Mathematics, 03.09.2020 21:01

Mathematics, 03.09.2020 21:01

Biology, 03.09.2020 21:01

History, 03.09.2020 21:01

Law, 03.09.2020 21:01


Mathematics, 03.09.2020 21:01

Mathematics, 03.09.2020 21:01


Mathematics, 03.09.2020 21:01






Mathematics, 03.09.2020 21:01


Business, 03.09.2020 21:01

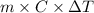
 = change in temperature
= change in temperature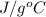 , water is 4.18
, water is 4.18 

