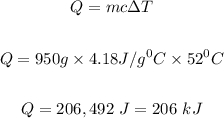Chemistry, 05.02.2023 09:40 geraldmorgan5580
How many kilojoules of heat are absorbed when 950 mL of water is heated from 27 degrees Celsius to 79 degrees Celsius? The specific heat of water is 4.18 J/g-C. Answer choices: 104 kJ 12 kJ 120 kJ 206 kJ

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which supports the idea that birds and butterflies both have wings but they do not have a common ancestor with wings? a. the wings are analogous structures that evolved differently and do not have a similar internal structure. b. the wings are homologous structures that evolved differently and do not have a similar internal structure. c. wings of birds are vestigial structures, but the wing structures of bats are not vestigial. d. wings of bats are vestigial structures, but the wing structures of birds are not vestigial
Answers: 1

Chemistry, 22.06.2019 13:00
What happens to the average kinetic energy of a gas when the particles of the gas collide against each other at a constant temperature and volume? explain your answer.
Answers: 3

Chemistry, 22.06.2019 14:00
650.j is the same amount of energy as? 2720cal1550cal650.cal2.72cal
Answers: 2

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 3
You know the right answer?
How many kilojoules of heat are absorbed when 950 mL of water is heated from 27 degrees Celsius to 7...
Questions


Chemistry, 03.03.2021 20:00

Law, 03.03.2021 20:00


Mathematics, 03.03.2021 20:00


Mathematics, 03.03.2021 20:00

Mathematics, 03.03.2021 20:00

Mathematics, 03.03.2021 20:00



Social Studies, 03.03.2021 20:00

Mathematics, 03.03.2021 20:00

Mathematics, 03.03.2021 20:00

History, 03.03.2021 20:00

Mathematics, 03.03.2021 20:00

Physics, 03.03.2021 20:00