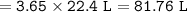Chemistry, 10.03.2022 14:40 NathanaelLopez
If one mol of gas = 22.4 at STP how many liters is occupied by 3.65 moles of oxygen gas at STP

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Asample of neon occupies a volume of 375 ml at stp. what will be the volume of neon if the pressure is reduced to 90.0 kpa? a. 422 ml b. 422 l c. 333 ml d. 333 l
Answers: 2

Chemistry, 22.06.2019 13:10
What type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? view available hint(s) what type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? salt bridge disulfide bridge hydrogen bond hydrophobic interaction
Answers: 1

Chemistry, 22.06.2019 16:10
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1

Chemistry, 22.06.2019 20:00
How are the terms group and period used on the periodic table
Answers: 1
You know the right answer?
If one mol of gas = 22.4 at STP how many liters is occupied by 3.65 moles of oxygen gas at STP...
Questions

Social Studies, 25.12.2021 09:20

Health, 25.12.2021 09:20



Health, 25.12.2021 09:20







Computers and Technology, 25.12.2021 09:20

Mathematics, 25.12.2021 09:20


SAT, 25.12.2021 09:20


History, 25.12.2021 09:30