
Chemistry, 03.03.2022 05:30 ALGEBRAHELP9504
Which would be the best way to represent the concentration of a 1. 75 M K2CrO4 solution? 1. 75% K2CrO4 [K2CrO4] (K2CrO4) K2CrO4, [M] = 1. 75.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 05:40
Consider the elements bromine and chlorine; which elements has a larger ionic radius ?
Answers: 1

Chemistry, 22.06.2019 07:30
Which of the following best supports the concept that genetic information is passed on to offspring from both of their parents, not just one?
Answers: 2

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2
You know the right answer?
Which would be the best way to represent the concentration of a 1. 75 M K2CrO4 solution? 1. 75% K2Cr...
Questions


History, 10.03.2021 01:00


Mathematics, 10.03.2021 01:00

Mathematics, 10.03.2021 01:00

Health, 10.03.2021 01:00



Mathematics, 10.03.2021 01:00


Mathematics, 10.03.2021 01:00

History, 10.03.2021 01:00


Mathematics, 10.03.2021 01:00

Mathematics, 10.03.2021 01:00

Mathematics, 10.03.2021 01:00


Mathematics, 10.03.2021 01:00

Mathematics, 10.03.2021 01:00

History, 10.03.2021 01:00

![\rm [K_{2}CrO_{4}].](/tpl/images/2676/1464/eb07c.png)
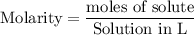
![\rm [X]](/tpl/images/2676/1464/76683.png) or
or ![\rm [K_{2}CrO_{4}]](/tpl/images/2676/1464/116cf.png) is the representation of the concentration.
is the representation of the concentration.

