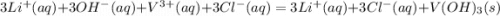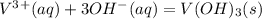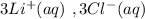Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:10
Select the correct answer from each drop-down menu.describe what happens to a carbon-11 atom when it undergoes positron emission.the decay of a carbon-11 atom _1_, and this causes it to emit _2_.options for 1: > changes a neutron into a proton> changes a proton into a neutron> is hit with a neutron> reconfigures its protons and neutronsoptions for 2: > a negatively charged electron-sized particle> a positively charged election-sized particle> two atoms and several neutrons> two neutrons and two protons
Answers: 3

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3


You know the right answer?
HELP ME WITH THIS PLEASE! WILL MARK BRAINLIEST!
Please be specific and label which one goes with e...
Questions

SAT, 20.10.2021 14:00

Business, 20.10.2021 14:00


Mathematics, 20.10.2021 14:00



Mathematics, 20.10.2021 14:00

Chemistry, 20.10.2021 14:00


Chemistry, 20.10.2021 14:00



Chemistry, 20.10.2021 14:00



Computers and Technology, 20.10.2021 14:00

Mathematics, 20.10.2021 14:00



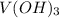 .
.