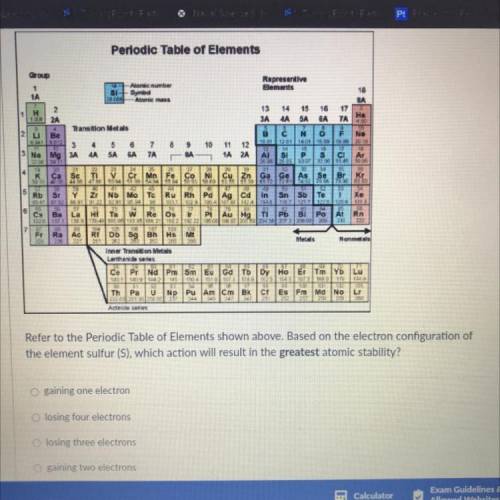
HELP PLEASE ASAP!!! GIVING OUT BRAINLIST
...

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2

Chemistry, 23.06.2019 06:00
Amanda pushes a box across the room with a force of 30 n. it accelerates at 5 m/s/s. what is the mass of the box? * 6 kg 1.16 kg 30 kg 5kg
Answers: 2

Chemistry, 23.06.2019 13:00
Me puede ayudar con estas preguntas 1. diga que estudia la química orgánica explica el nacimiento bioquímicas 2. cual es la importancia de la química orgánica. 3. determine las principales características del hidrógeno, oxigeno, nitrógeno y azufre como elementos que constituyen los compuestos orgánicas. 4. elabore una tabla comparativa entre compuestas orgánicas e incaicos.
Answers: 1

Chemistry, 23.06.2019 13:30
Explain the impact that changing the temperature has on a system in a state of dynamic equilibrium. what will happen when the temperature of an exothermic reaction mixture at equilibrium is increased?
Answers: 3
You know the right answer?
Questions

Computers and Technology, 23.10.2020 16:40

Mathematics, 23.10.2020 16:40

History, 23.10.2020 16:40


Chemistry, 23.10.2020 16:40



English, 23.10.2020 16:40


English, 23.10.2020 16:40



Chemistry, 23.10.2020 16:40



Mathematics, 23.10.2020 16:40


Mathematics, 23.10.2020 16:40


Health, 23.10.2020 16:40