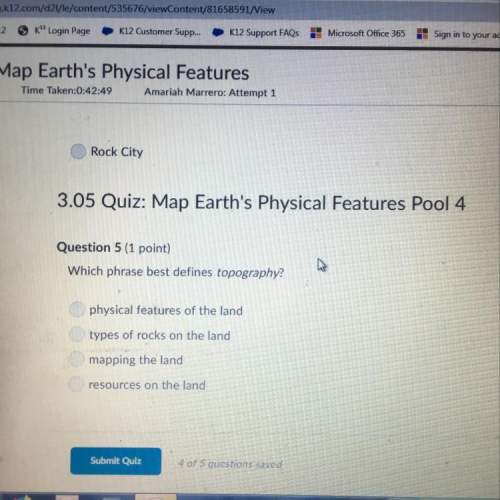
Chemistry, 17.12.2021 01:20 milagrosee12
VERY URGENT! IB CHEMISTRY
0.0810 g of a group 2 metal iodide, MI2, was dissolved in water and made up to a total volume of 25.00 cm3.
Excess lead(II) nitrate solution (Pb(NO3)2(aq)) was added to the MI2 solution to form a precipitate of lead(II)
iodide (PbI2). The precipitate was dried and weighed and it was found that 0.1270 g of precipitate was obtained.
a Determine the number of moles of lead iodide formed.
b Write an equation for the reaction that occurs.
c Determine the number of moles of MI2 that reacted.
d Determine the identity of the metal, M.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
The agent of mechanical weathering in which rock is worn away by the grinding action of other rock particles is call
Answers: 1


Chemistry, 22.06.2019 20:30
Calculate the percent composition by mass of each element in al(oh)3. use at least three significant figures.
Answers: 1

Chemistry, 23.06.2019 01:30
What is the importance of interlocking the fingers and rubbing while washing hands? the palms are the dirtiest parts of the hands. the spaces between the fingers get washed. the backs of the hands get washed. the fingernails are the dirtiest parts of the hands
Answers: 1
You know the right answer?
VERY URGENT! IB CHEMISTRY
0.0810 g of a group 2 metal iodide, MI2, was dissolved in water and made...
Questions

English, 25.11.2021 15:20

Mathematics, 25.11.2021 15:20

Chemistry, 25.11.2021 15:20

Computers and Technology, 25.11.2021 15:20


Computers and Technology, 25.11.2021 15:20

Mathematics, 25.11.2021 15:20


English, 25.11.2021 15:20

Mathematics, 25.11.2021 15:20


History, 25.11.2021 15:20


Physics, 25.11.2021 15:20

Mathematics, 25.11.2021 15:20

Computers and Technology, 25.11.2021 15:20




Biology, 25.11.2021 15:20