Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 23.06.2019 02:30
Asubstance is held in an open container. its particles move past one another at random speeds but do not leave the container. heat is removed from the system, and the particles slow down. when enough heat is removed, the particles no longer have enough speed to overcome the weak attractive forces between them. when this happens, the substance enters its solid state. the process described above is known as .
Answers: 3


Chemistry, 23.06.2019 12:10
Which structure is a valid representation of a hydrocarbon molecule?
Answers: 2
You know the right answer?
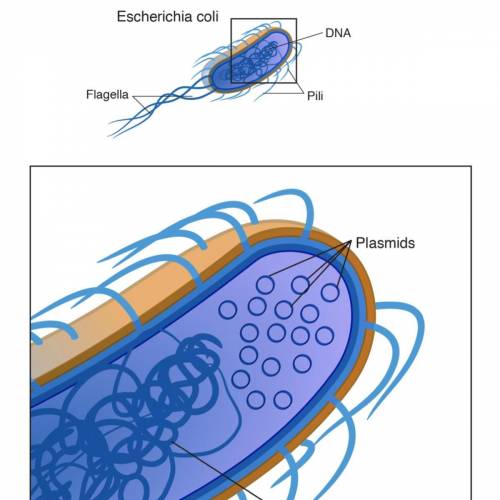
Do plasmids have an importance beyond the practice of genetic engineering?...
Questions

Mathematics, 26.05.2021 01:00


English, 26.05.2021 01:00

History, 26.05.2021 01:00



Health, 26.05.2021 01:00

Mathematics, 26.05.2021 01:00

Mathematics, 26.05.2021 01:00

Mathematics, 26.05.2021 01:00

Mathematics, 26.05.2021 01:00

History, 26.05.2021 01:00


Chemistry, 26.05.2021 01:00


Biology, 26.05.2021 01:00



Mathematics, 26.05.2021 01:00

History, 26.05.2021 01:00