Chemistry, 13.12.2021 07:10 heybrothwrlogan
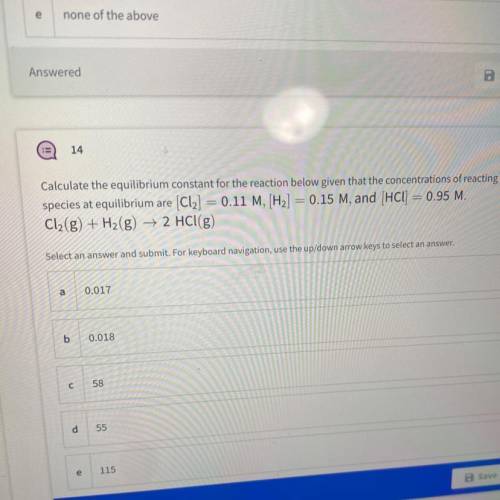
Calculate the equilibrium constant for the reaction below given that the concentrations of reacting
species at equilibrium are (Cl2] = 0.11 M, [H2] = 0.15 M, and (HCl) = 0.95 M.
Cl2(g) + H2(g) → 2 HCl(g)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
As you move from right to left on the periodic table the atomic radius fill in the blank
Answers: 2

Chemistry, 22.06.2019 09:50
Although respiratory organs vary across different organisms, they all contain respiratory surfaces that have a large surface area and are extremely thin. explain why having an extremely thin respiratory surface with a large surface area is advantageous for the process of gas exchange
Answers: 1

Chemistry, 22.06.2019 17:30
I'm learning about the periodic tables and what each subject's configuration is. for example, hydrogen is 1s^1, but i don't understand how you get that. can someone me understand how to figure out how to figure this out? sorry if the question makes no sense, but it would really a lot if you could me understand! you so much if you can!
Answers: 1

Chemistry, 23.06.2019 04:10
What does the field of thermodynamics relate to a-changes in nuclear reactions b- changes in energy in systems c changes in molecular structure d changes in atomic properties
Answers: 1
You know the right answer?
Calculate the equilibrium constant for the reaction below given that the concentrations of reacting...
Questions



History, 26.05.2020 03:58

Mathematics, 26.05.2020 03:58

Business, 26.05.2020 03:58

Mathematics, 26.05.2020 03:58

Mathematics, 26.05.2020 03:58

Mathematics, 26.05.2020 03:58

Mathematics, 26.05.2020 03:58


Mathematics, 26.05.2020 03:58


Biology, 26.05.2020 03:58

Mathematics, 26.05.2020 03:58

History, 26.05.2020 03:58

History, 26.05.2020 03:58

Spanish, 26.05.2020 03:58


Arts, 26.05.2020 03:58